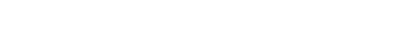Glycoproteins in Cancer Diseases
Posted in Projects
Cancer diseases are not typically associated with mutations in the glycosylation pathways but glycosylation is frequently altered in their progression. We study most actively the progression of fibrotic liver disease of various etiologies to hepatocellular carcinoma (HCC). Quantification of the glycoforms of liver secreted glycoproteins is an avenue for the identification of disease markers used in non-invasive serologic disease monitoring. We find that fucosylation od N-glycans increases at all stages of the fibrotic disease both at the core and the outer arms. Branching of the glycans also increases and core fucosylation of the branched N-glycans provides a sensitive readout of the fibrotic liver disease.
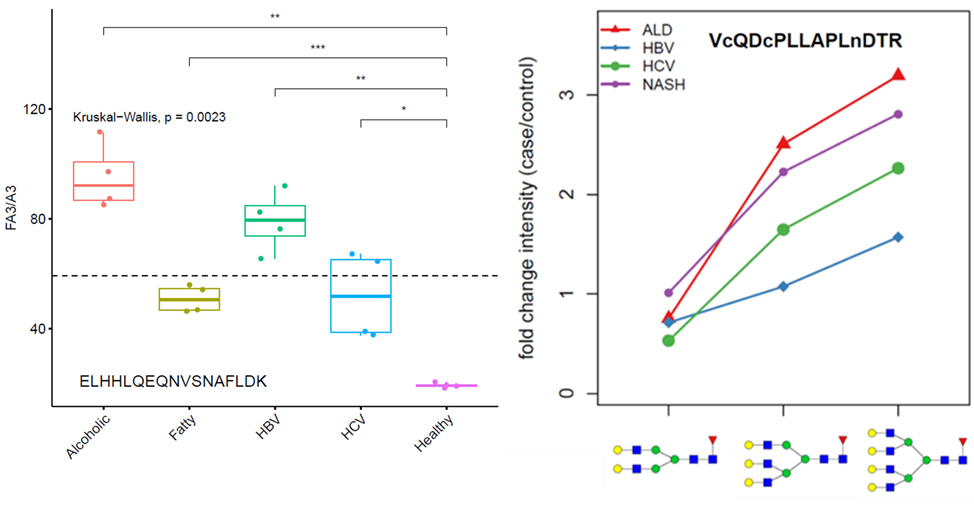
Figure 1. Core fucosylated glycopeptides on branched structures provide a sensitive serologic readout of the progression of fibrotic liver disease.
Fibrosis of the liver is further associated with altered mucin type O-glycosylation and increased sialylation of the T-antigen on specific glycoproteins further improves non-invasive serologic monitoring of the disease.
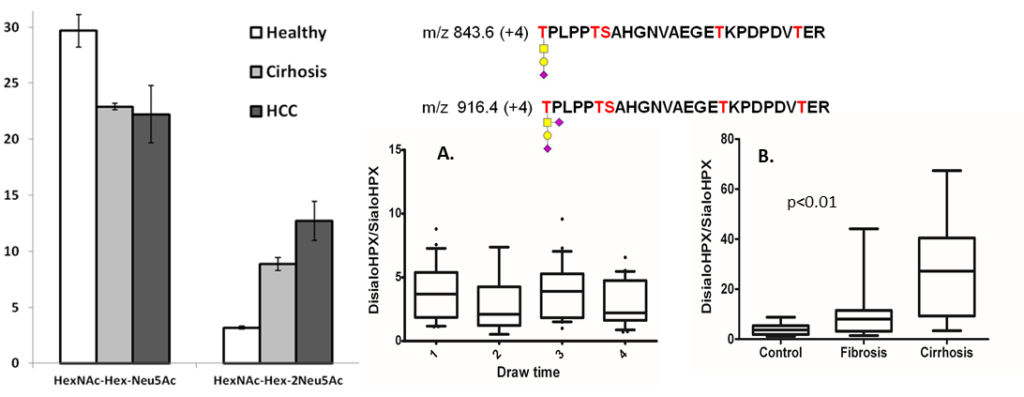
Figure 2. Sialylated glycoforms of hemopexin increase in the serum of patients at progressive stages of liver fibrosis. Sanda et al (2016) Clin Prot. 21, 13-24.
The availability of data on genomic polymorphisms, mutations, gene expression, or protein quantities from recent cancer proteogenomic studies allow integration of the protein glycosylation pathways into our studies of the interactions of the transformed cells with their host environment.
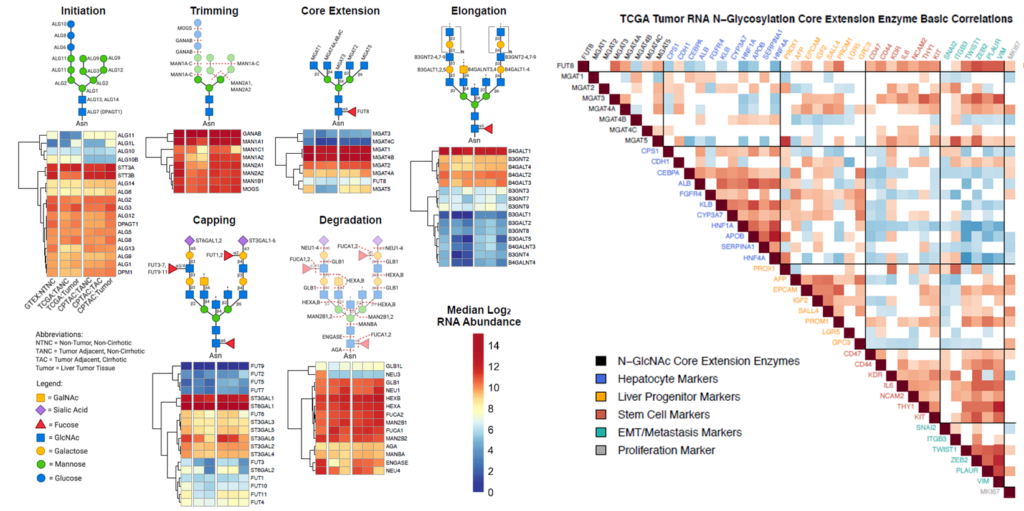
Figure 3. Association of glycosylation pathways with phenotypic characteristics of cancer diseases.
Identification of the glycoprotein targets and their functions reveals novel options for disease monitoring and therapeutic interventions.